Evaluation of the Tocilizumab therapy in human cancers: Latest evidence and clinical potential - Ghasemi - 2022 - Journal of Clinical Pharmacy and Therapeutics - Wiley Online Library

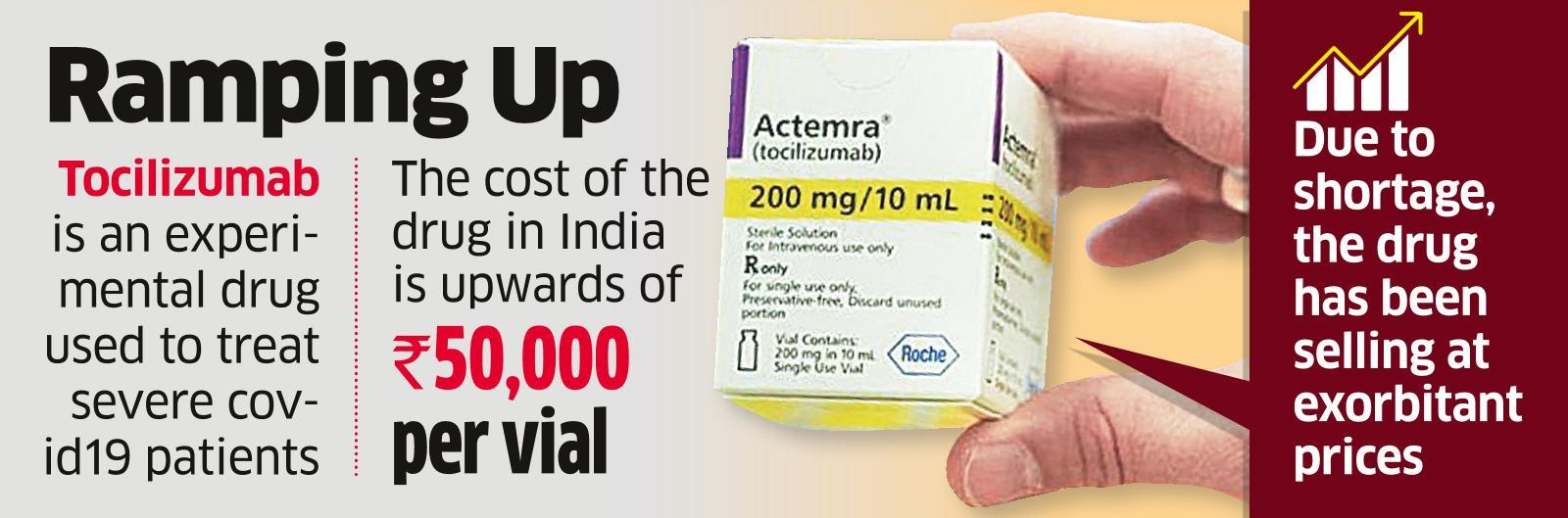
Tocilizumab Injection Supply: Delhi HC asks Centre to hold meeting with Roche to ensure immediate supply of Tocilizumab - The Economic Times
USTC Global on X: "The mechanism of using Tocilizumab to cure severe patients of COVID-19. @NIH @CDC @DrAiLynTan #coronavirus #COVID19 #CoronavirusOutbreak #coronaviruskorea #ItalyCoronavirus #italylockdown #IranCoVidTruth #Tocilizumab https://t.co ...
Approval lapsed) RoActemra tocilizumab 80mg/4mL concentrate for solution for infusion (Roche Germany) | Therapeutic Goods Administration (TGA)
Approval lapsed) RoActemra tocilizumab 200 mg/ 10mL concentrate for solution for infusion (Roche Germany) | Therapeutic Goods Administration (TGA)
Roche Suspends Patents On Tocilizumab In LMICs After WHO Recommends It As Treatment For Severe COVID-19 - Health Policy Watch
Sickest coronavirus patients to be treated with arthritis drugs after study finds reduction in death rates - ABC News
Approval lapsed) RoActemra tocilizumab 400 mg/ 20mL concentrate for solution for infusion (Roche Germany) | Therapeutic Goods Administration (TGA)






/cloudfront-us-east-2.images.arcpublishing.com/reuters/QFWUTH5BLZN5LGWVT27LGWPZBE.jpg)